As both elements have undergone a change in their oxidation state, we can identify the reaction as a redox reaction. Chapters Reactivity Series of Metals - GCSE Chemistry KayScience 17.9K subscribers Subscribe 9.2K views 2 years ago In this video you will learn all the science for this. Chlorine has gained 1 electron to gain a -1 charge. After the reaction, sodium has lost 1 electron to gain a +1 charge. Much like balancing a reaction, we can tally the oxidation states of the reactants and products to see if there has been a change in oxidation state:īoth reactants start out with an oxidation state of 0 (i.e. In this process (known as smelting) iron (III) oxide is heated to a very high temperature (above 1200\degree \text One common example of this method is the the reduction of iron (III) oxide to produce pure iron. Author: Kelly Ryder Created Date: 05:32:00 Title: Reactivity series of metals worksheets follow-up Description: Supports the RSC practical video Reactivity series of metals Keywords. Reduction by carbon is a preferable method of extraction, as this is much less expensive than extraction using electrolysis. Reactivity series of metals Supporting resources. This is because metal oxides will not lose their oxygen when the metal is more reactive than carbon. Those lower in the reactivity series and so are less reactive will be reduced by reaction with carbon. Metals that come higher in the reactivity series and so are more reactive than carbon will be reduced using electrolysis. The easier it is for a metal to form its positive ion, the more reactive. Metals high in the reactivity series can displace metals lower down. this technique avoids traditional mining methods of digging, moving and disposing of large amounts of rock. When metals react with other substances, the metal atoms always form positive ions. GCSE CHEMISTRY METALS & THE REACTIVITY SERIES. Which method is used will depend on the position of the metal in the reactivity series relative to that of carbon. Metals can be arranged in order of how reactive they are. YUMPU automatically turns print PDFs into web optimized ePapers that Google loves. This is typically done in one of two ways: 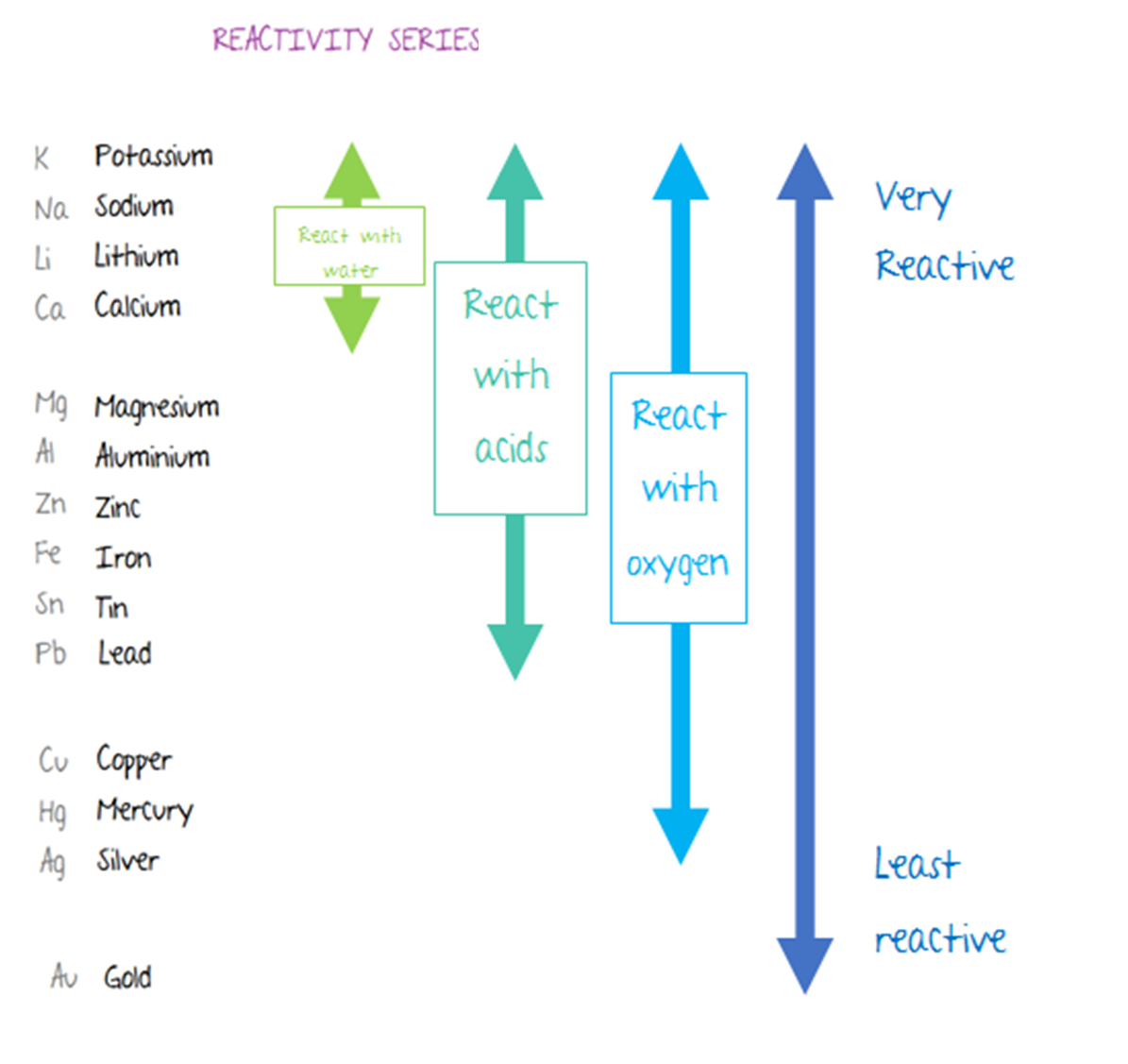
However, copper can be extracted using carbon or hydrogen.To extract pure metals from their oxide ores, they must be reduced (i.e. 
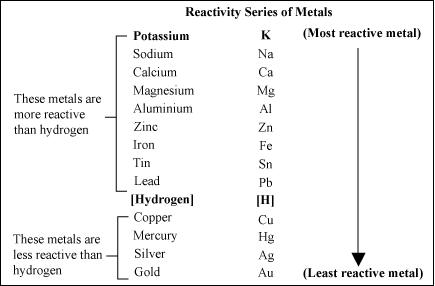
Note that zinc and iron can be displaced from their oxides using carbon but not using hydrogen. Here is the reactivity series including carbon and hydrogen: It is useful to place carbon and hydrogen into the reactivity series because these elements can be used to extract metals. When this layer is removed, the observations are more reliable. This is because its protective aluminium oxide layer makes it appear to be less reactive than it really is. Note that aluminium can be difficult to place in the correct position in the reactivity series during these experiments. The quicker the fizzing, the more reactive the metal. The speed at which hydrogen bubbles are produced tells us how reactive a metal is with acid. The tables show how the elements react with water and dilute acids: Element Iron and aluminium are extracted from their ores in. The reactivity of a metal is related to its tendency to form positive ions. Observations of the way that these elements react with water, acids and steam enable us to put them into this series. The reactivity series shows metals in order of reactivity. For example: P eople S ay L ittle C hildren M ake A Z ebra I ll C onstantly S niffing G iraffes. The reactivity series of metalsĪ good way to remember the order of a reactivity series of metals is to use the first letter of each one to make up a silly sentence. More reactive metals have a greater tendency to lose electrons and form positive ions. displacement reaction occurs there is a temperature rise. Magnesium(More reactive metal) + Zinc sulphate (Less reactive salt ). 1 (a) The reactivity of metals can be studied using displacement reactions. In a reactivity series, the most reactive element is placed at the top and the least reactive element at the bottom. More reactive metal will displace the less reactive metal from its salt solution. The order of reactivity is Potassium, sodium, calcium, magnesium, aluminium, carbon, zinc, iron, tin, lead, hydrogen, copper, silver, gold, platinum. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
